Introduction
What makes a healthy soil? Is soil merely a solid medium that holds nutrients for plant growth? Increasing concern for the sustainability of our natural resources has led to the development of a more complex concept of soil health. Karlen et al. (1997) proposed the following as vital soil functions: (1) sustaining biological activity, diversity, and productivity; (2) regulating and partitioning water and solute flow; (3) filtering, buffering, degrading, immobilizing, and detoxifying organic and inorganic materials, including agricultural, industrial and municipal by-products and atmospheric deposition; (4) storing and cycling nutrients and other elements within the Earth's biosphere; and (5) providing support of socioeconomic structures and protection for archeological treasures associated with human habitation.
The term "soil quality" has been coined to describe the combination of chemical, physical, and biological characteristics that enables soils to perform a wide range of functions. We have described in this publication: (1) some indicators of soil that can be measured with a simple test kit developed by the United States Department of Agriculture, Natural Resource Conservation Service (USDA-NRCS); (2) directions for interpreting these measurements; (3) the effects of soil amendments on soil quality attributes; and (4) comparisons of field kit and laboratory results.
Quantifying Soil Quality Indicators
The assessment of soil quality requires quantification of critical soil attributes. Initial measurements of soil quality attributes should be made in high and low productivity areas to establish ranges of values that are site specific. Changes occurring over time can then be measured to evaluate effects of different practices. Detailed information on when, where, why, and how to measure soil quality is presented in the Soil Quality Testing Kit Instruction Manual and the Soil Quality Interpretation Guide published on the USDA-NRCS - Soil Quality Institute website (see the "Resources" section of this document for the address). Descriptions of soil biological, physical and chemical indicators follow.
Soil Biological Indicators
Soil microorganisms (fungi and bacteria) and other fauna (e.g., earthworms, insects, arthropods) influence the availability of nutrients for crop growth by decomposing soil organic matter and releasing or immobilizing plant nutrients. Biological activity improves soil aggregation through the secretion of soil binding mucilages and hyphal growth. Improved aggregation, in turn, increases water infiltration and the ease of plant root penetration. Soil biological activity is considered an integral attribute of a healthy soil.
Soil respiration rate
Soil respiration rate [as assessed by carbon dioxide (CO2) evolution] is an indicator of soil biological activity. Soil CO2 evolution results from the decomposition of organic matter; thus, soil respiration rate is an indicator of the amount of decomposition that is occurring at a given time.
Soil respiration can be limited by moisture, temperature, oxygen, soil reaction (i.e., pH), and the availability of decomposable organic substrates. Optimum respiration usually occurs around 60% of water filled pore space. Soil respiration will decrease under saturated or dry conditions. Biological activity doubles for every 18°F rise in temperature until the optimum temperature is reached (varies for different organisms). Activity declines as temperature rises above optimum. The most efficient soil organic matter decomposers are aerobic; thus, soil respiration rates decline as soil oxygen concentration decreases. Oxygen is most limiting in soils that are saturated with water. Greater oxygen flow occurs in well-aggregated soils that have many macropores.
Addition of organic materials will generally increase soil respiration. Organic matter provides the food or substrate on which heterotrophic soil microbes feed. Organic materials with low carbon to nitrogen (C:N) ratios (e.g. manure, leguminous cover-crops) are easily decomposed; thus, the addition of these materials to soil will increase soil respiration. Materials with high C:N ratios (e.g., compost, sawdust) decompose more slowly but provide a more stable, long term supply of organic material than legumes, biosolids, and manures. Soil microbes will compete with plants for nitrogen when soil is amended with products having C:N ratios higher than 25:1.
Agricultural chemicals that directly kill or otherwise impair soil microorganisms, such as fungicides and nematocides, reduce soil respiration. Although these chemicals target pathogenic organisms, they may also impair the viability of beneficial organisms.
Organic matter decomposition provides benefits and drawbacks. Decomposition of organic matter is the primary route through which some essential nutrients (e.g., nitrogen) are released, but organic matter destruction reduces the benefits that organic matter confers to soil physical and chemical properties. The addition of organic materials to the soil must equal the loss due to decomposition for the sustainability of the system to be maintained.
| Management factors influencing soil respiration |
| Increases soil respiration | | Decreases soil respiration |
- Adding organic amendments, such as manure, biosolids, and crop residues
- Irrigating to proper moisture content
| |
- Removing or burning crop residues
- Continuous tillage
- Agricultural chemicals (e.g., fungicides and nematocides)
|
Interpretation of soil respiration values
In general, a higher respiration rate indicates better soil quality. Low respiration rate, when soil temperature and moisture are favorable for biological activity, would indicate less than desirable organic matter input. This value must be interpreted within the context of other indicators. For example, a very low nitrate concentration plus a high respiration rate may indicate a high nitrogen immobilization rate, possibly due to the addition of crop residues or other soil amendments that possess wide C:N ratios. Some general guidelines to interpreting respiration values are presented in Table 1. These are only guidelines and should not be applied to every soil type and management situation.
| Table 1. General soil respiration class ratings and soil condition at optimum soil temperature and moisture conditions, primarily for agri-cultural land uses (Woods End Research, 1997). |
Soil respiration
(lbs. CO2 -C/ac/day) | Class | Soil condition |
| 0 | No soil activity | Soil has no biological activity and is virtually sterile. |
| < 9.5 | Very low soil activity | Soil is very depleted of available organic matter and has little biological |
| 9.5 - 16 | Moderately low soil activity. | Soil is somewhat depleted of available organic matter, and biological activity is low. |
| 16 - 32 | Medium soil activity | Soil is approaching or declining from an ideal state of biological activity. |
| 32 - 64 | Ideal soil activity | Soil is in an ideal state of biological activity and has adequate organic matter and active populations of microorganisms. |
| > 64 | Unusually high soil activity | Soil has a very high level of microbial activity and has high levels of available organic matter, possibly from the addition of large quantities of fresh organic matter or manure. |
Soil Physical Indicators
The physical properties of soils - texture, structure, density, porosity, water content, strength, temperature, and color - determine the availability of oxygen in soils, the mobility of water into or through soils, and the ease of root penetration. Some of these properties are immutable (e.g., texture) and cannot be modified by cultural practices, but density, water holding capacity, and porosity can be improved using appropriate soil management techniques.
Soil bulk density
Soil bulk density is the mass of soil per unit volume in its natural field state and includes air space and mineral plus organic materials. Bulk density gives useful information in assessing the potential for leaching of nutrients, erosion, and crop productivity. Runoff and erosion losses of soil and nutrients can be caused by excessive bulk density when surface water is restricted from moving through the soil. Bulk density provides an estimate of total water storage capacity when the soil moisture content is known.
| Management factors influencing bulk density |
| Increases bulk density | | Decreases bulk density |
- Continuous tillage
- Removing or burning residue
| |
- Continuous cropping
- Adding organic amendments
- Trafficking on wet soils
|
Interpretation of bulk density measurements
Bulk density can be used to determine if soil layers are too compact to allow root penetration or adequate aeration. Bulk densities that limit plant growth vary for soils of different textural classes (Table 2) (Arshad et al., 1996).
| Table 2. General relationship of soil bulk density to root growth based on soil texture. |
| Soil texture | Ideal bulk densities
(g/cm3) | Bulk densities that may affect root growth
(g/cm3) | Bulk densities that restrict root growth
(g/cm3) |
| Sands, loamy sands | <1.60 | 1.69 | >1.80 |
| Sandy loams, loams | <1.40 | 1.63 | >1.80 |
| Sandy clay loams, clay loams | <1.40 | 1.60 | >1.75 |
| Silts, silt loams | <1.30 | 1.60 | >1.75 |
| Silt loams, silty clay loams | <1.40 | 1.55 | >1.65 |
| Sandy clays, silty clays, some clay loams (35-45% clay) | <1.10 | 1.49 | >1.58 |
| Clays (>45% clay) | <1.10 | 1.39 | >1.47 |
Water Infiltration
Infiltration is the process of water entering the soil. Infiltration rate is dependent on the soil type; soil structure, or amount of aggregation; and the soil water content (Lowery et al., 1996). Soil crusting, compaction, vegetative cover, and root and earthworm channels also influence the ability of water to move through (infiltrate) soil layers. Infiltration is important for storing water in the soil profile for plant growth and for reducing runoff and erosion. Infiltration rates are best determined when the soil is at or near field capacity, usually12 to 48 hours after the soil has been thoroughly wetted (i.e., soaking rain or irrigation).
Management practices that affect soil crusting and compaction, vegetative cover, and soil porosity will increase or decrease the rate of water infiltration. For example, slow infiltration can be caused by increased soil compaction or loss of surface soil structure (reduced aggregation) through tillage. Leaving crop residues on the soil surface or increasing the organic matter content in the soil surface may maintain aggregation and enhance infiltration
| Management factors influencing infiltration rates |
| Increases infiltration rate | | Decreases infiltration rate |
- Adding organic amendments
- Continuous cropping
| |
- Removal or burning crop residue
- Continuous tillage
- Compaction as a result of traffic
|
Interpretation of infiltration rates
The infiltration rate is most responsive to conditions near the soil surface and changes drastically with management (Sarrantonio et al., 1996). Infiltration is rapid into large continuous pores at the soil surface and decreases as the size of these pores is reduced. Some general values for infiltration into soils of varying textural classes are presented in Table 3. These are average values and should not be generalized for all soil types.
| Table 3. Steady infiltration rates for general soil texture groups in very deeply wetted soil (Hillel, 1982). |
| Soil type | | Steady infiltration rate
(inches per hour) |
| Sands | | >0.8 |
| Sandy and silty soils | | 0.4-0.8 |
| Loams | | 0.2-0.4 |
| Clayey soils | | 0.04-0.2 |
| Sodic clayey soils | | <0.04 |
Field water-holding capacity
Field water-holding capacity is the amount of water a soil can hold after being saturated and allowed to drain for a period of one to two days. Field water-holding capacity is influenced by soil texture (relative amount of silt-, clay-, and sand-sized particles), aggregation, organic matter content, and overall soil structure.
| Management factors influencing field water-holding capacity (WHC) |
| Increases field WHC | | Decreases field WHC |
- Adding organic amendments crop residue
- Conservation tillage
- Cover cropping
| |
- Removing and burning
- Conventional tillage
|
Interpretation of field water-holding capacity
Generally, a soil with a high water-holding capacity will provide more plant-available water, but soil texture also determines what portion of the soil water is available to plants. Clayey soils hold the most, sandy soils the least, and loamy soils intermediate amounts of water; however, loamy soils provide the most plant available water because much of the water in the small pores in clayey soils is held too tightly to be available to plants
(Figure 1). Organic matter physically holds more water than does mineral matter; thus, increasing a soil's organic matter content increases its water-holding capacity. The major management practices that influence water-holding capacity are tillage and crop residue management. Soils that are highly tilled tend to lose water-holding capacity. Tillage reduces the content of organic matter and reduces pore volume.
Soil Chemical Properties
Soil chemical properties are determined by the amounts and types of soil colloids (clays and organic matter). Chemical properties include mineral solubility, nutrient availability, soil reaction (pH), cation exchange capacity, and buffering action.
Soil pH
Soil pH indicates how acid (pH<7, high H+ concentration) or basic (pH>7, high OH- concentration) is the soil solution. Soil pH is influenced by parent soil materials and tends to decrease with time. Soils with low base (Ca, Mg, K, etc.) status, such as those in the Southeastern United States, are sensitive to the acidifying effects of nitrogen fertilizers (including organic N sources). The addition of limestone and other basic materials is normally used to maintain soil pH in a desirable range. Although organic matter additions may not directly affect soil pH, soils that receive significant amounts of organic materials tend to maintain (buffer) soil pH values for longer periods of time.
Interpretation of Soil pH
Most crops grown in the temperate mid-Atlantic and southeastern United States prefer slightly acidic soils (i.e., pH of 5.6 to 6.8). Lower or higher pH values can cause plant nutrient deficiencies (e.g., P, Mn, Zn, Cu, Fe, Mo) or elemental toxicities (i.e., Al, Mn), which have adverse effects on crop yield (Figure 2).
Soil Nitrate
Nitrate (NO3-) and ammonium (NH4+) are the only forms of nitrogen that can be used by plants. Chemical nitrogen fertilizers add ammonium or nitrate directly to soil. The organically complexed forms of nitrogen in crop residues, compost, and manure must be mineralized into inorganic ammonium before they can be used by plants. Ammonium is either directly taken up by plant roots or soil microorganisms, or converted to nitrate. Most inorganic N in agricultural soils is taken up by crops in the nitrate form of nitrogen.
Nitrate can leach through soil if it is not taken up by plant roots or soil microorganisms. Nitrate that is leached through the soil profile may contaminate surface water and groundwater and is unavailable for plant uptake. The goal in managing nitrogen in the soil should be to balance crop needs with supply. This will reduce pollution potential and maintain economically viable crop yields.
Interpretation of nitrate values
The nitrogen cycle is so complex that it is difficult to predict the amount of available N from various forms of the nutrient in soil. Soil nitrate analysis may be used to compare the available N from different soil amendments and the effects of soil management on nitrate leaching potential. Nitrate concentration in the top 12 inches of soil at the 10 to 12 inch stage of corn growth has been used successfully to predict the sufficiency of N for the crop (Evanylo and Alley, 1996; Evanylo and Alley, 1997); however, further calibration will be necessary to develop soil nitrate sufficiency norms for other crops.
The effect of organic amendments on soil quality indicators and comparison of field and laboratory methods
Soil amendments vary in their effects on soil properties. Organic amendments maintain soil aggregate stability and contribute to a soil's water holding capacity, while inorganic amendments, such as commercial fertilizer, do not benefit soil physical properties. Furthermore, differences exist in the quality of organic matter. More stable organic amendments may have more lasting effects on soil physical properties, but may not be as easily assimilated by soil organisms as less decomposed materials. A study was designed to demonstrate the effects of incorporated inorganic chemical fertilizer, cover crop, cotton gin trash compost and swine manure on soil quality. The specific objectives were:
- To compare the results of the soil quality field test kit with standard laboratory procedures for the same tests, and
- To determine if the soil quality indicators measured by the soil quality field test kit are responsive to various soil amendment treatments.
Methods and Materials
Soil testing was performed on research plots at the Center for Environmental Farming Systems in Goldsboro, NC, on June 3-4, 1998. These plots had been amended twice in a 16-month period with agronomic rates (i.e., the amount estimated to supply the crop N needs of 120 lbs N per acre) of inorganic fertilizer, composted cotton gin trash (CGT), and swine manure, or an incorporated cover crop residue. Each treatment was replicated four times. All plots were roto-tilled and hilled prior to transplanting tomato (Lycopersicon esculentum Mill.) for the second consecutive season. No tillage was performed during the growing season and surface mulching with straw was used for weed control.
Each plot was sampled during the growing season at one location on the hilled portion for assessment of soil respiration (CO2 evolution), water infiltration, bulk density, soil pH, soil nitrate, and field water-holding capacity. Bulk density, soil pH, soil nitrate, and field water-holding capacity were each analyzed by standard laboratory and field kit. Soil respiration and water infiltration were only measured with the field kit. Laboratory analyses were conducted by the following methods: bulk density (Blake, 1965), field water-holding capacity (Klute, 1965), pH (McLean, 1982), and nitrate (Keeney and Nelson, 1982; Lachat QuickChem methods No. 12-107-04-1-B, Zellweger Analytics, Inc., 6645 West Mill Road, Milwaukee, WI 53218-1239). For detailed descriptions of the materials and methods used for the field soil quality test kit, consult the soil quality handbook on the NRCS website: (http://www.statlab.iastate.edu:80/survey/SQI/sqihome.shtml) or Sarrantonio et. al. (1996).
Results
High air temperatures (e.g., 94°F on the sampling date) and below average rainfall for May and June 1998, resulted in high soil temperatures (83°F at 4 to 6 inch depth) and low available soil moisture at the time of sampling.
Soil respiration
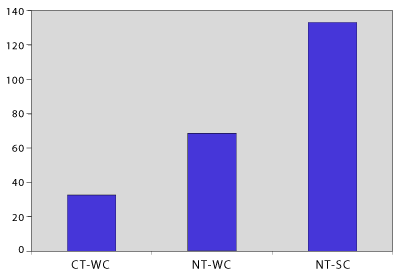
The soil respiration measurements under actual field temperature and moisture conditions were not statistically different among treatments. The data from the post wetting respiration test (after water saturation and draining overnight) was less variable and provided the best reflection of the trend of treatment differences because differences were more likely to be due to treatments than soil water status (Figure 3). Higher CO2 evolution occurred with amendments that provided greater amounts of easily decomposable (volatile) organic matter. Respiration response followed the pattern of manure > CGT > cover crops > inorganic fertilizer, although the respiration rate for cotton gin trash compost-amended soil was not significantly different than any soil. Carbon availability of microbially-stabilized plant-based compost is less than uncomposted manure and similar to weathered cover crop residue.
Water infiltration
Water infiltration rate was highest with the composted cotton gin trash compost as demonstrated by the shortest time for the water volume to infiltrate the soil (Figure 4). Infiltration rates for the other treatments were equal and somewhat slower than the cotton gin trash compost. The high organic matter content (~45%) of the compost probably increased the surface soil aggregation, prevented crusting, and maintained soil macropore flow more than the other treatments.
Density
Bulk density was lowest in the cotton gin trash compost amended soil, but no other treatments were different (Figure 5). The soil quality test kit bulk density test did not correlate well with the standard test for bulk density because we sampled soil from 3 to 6 inches with the standard core method and from 0 to 3 inches, where treatment effects were more apparent, with the field test kit. Another factor contributing to bulk density differences may have been the lower variability in the greater volume of soil sampled by the quick test kit than with the standard method. In this case, the test kit outperformed the standard test in assessing soil quality differences.
Soil pH
The lab and field pH measurements were positively correlated, but the field measurements were between 0.5 and 1.0 pH units higher than the lab (Figure 6). The large difference between the lab and the field pH measurements makes the field pH meter a poor choice for assessing soil pH. There were no treatment effects on soil pH with either the laboratory or the field kit measurements.
Soil Nitrate
Laboratory and field soil nitrate concentrations responded similarly to treatments, but nitrate concentrations were lower when measured by the field test kit. Soil nitrate was highest with the composted cotton gin trash and the swine manure and was lowest with the cover crop and inorganic fertilizer (Figure 7). The organic N sources were more effective than the inorganic fertilizer in maintaining soil nitrate concentrations in the root zone.
Field Water-Holding Capacity
Field water-holding capacity values were different when assessed by both methods, but the variability was less with the standard laboratory procedure (Figure 8). The general trend for both the test kit and the lab method was cotton gin trash > manure > cover crop > inorganic fertilizer. The most stabilized organic material (composted cotton gin trash) increased field moisture capacity more than inorganic fertilizer.
Differences among procedures probably occurred because: 1) the field kit measured the moisture content of an intact core, while the lab method measured the moisture content of a soil sample that was air-dried and sieved, which destroyed natural aggregation; and 2) the field kit employed actual field conditions to simulate water movement and holding capacity, whereas, lab method imposed an artificial pressure of -1/3 bar to simulate gravitational forces.
Conclusions and evaluation of the soil quality test kit
The test kit procedures are simple to understand and can be performed rapidly compared to standard laboratory analyses. A complete analysis of one sample takes approximately 30 minutes for the first day and 30 minutes to complete the second day's respiration measurement and water-holding capacity sampling. Two or more samples will take less than double or triple the time because analysis starting times can be staggered. The kit may have other on-farm applications besides soil testing. For example, the nitrate test can be used to monitor water quality.
The soil quality test kit results correlated well with standard laboratory analyses, although field measurements of pH were higher than acceptable. The field kit distinguished between soil management treatments in the field. The post-wetting or second day respiration test was more indicative of treatment differences because the variability due to soil moisture levels was reduced.
In general, organic amendments improved soil physical properties (infiltration rate, water-holding capacity, and bulk density) and increased biological activity (respiration rate) more than the inorganic commercial fertilizer. The organic amendments also maintained the highest concentrations of nitrate-N in the topsoil despite the higher application rate of readily plant-available nitrogen from commercial fertilizer than from the other amendments. The slow-release nature of the organic N probably prevented leaching losses of the same magnitude as from the inorganic fertilizer. Soil infiltration rate, water-holding capacity, bulk density, and nitrate-N were increased by the organic amendments in the order of their expected carbon stability (i.e., cover crop < manure < cotton gin trash compost), while respiration rate was highest with the manure.
The test kit should be used to identify the effects of management practices on general trends in soil quality rather than to measure absolute soil property values. Farmers, consultants, and educators can assess changes in soil properties to allow for the selection of management practices that can best enhance soil quality. The kit is an excellent tool for teaching the concepts of soil quality and is appropriate for use by Extension specialists (esp., Agriculture and Natural Resources, and 4-H) and other educators.
References
Arshad, M.A., B. Lowery, and B. Grossman. 1996. Physical tests for monitoring soil quality. p.123-142. In: J.W. Doran and A.J. Jones (eds.) Methods for assessing soil quality. Soil Sci. Soc. Am. Spec. Publ. 49. SSSA, Madison, WI.
Blake, G. R. 1965. In: C.A. Black (ed.) Methods of Soil Analysis, Part 1. Physical and Mineralogical Properties, Including statistics of Measurement and Sampling. ASA-SSSA, Agronomy Monograph 9: 374-390.
Doran J.W., D.C. Coleman, D.F. Bezdicek, and B.A. Stewart editors. 1994. Defining Soil Quality for a Sustainable Environment. SSSA Publ. No.35. Soil Science Society of America, 677 South Segoe Rd., Madison, WI 53711, USA.
Doran J.W., A.J. Jones editors. 1996. Methods for Assessing Soil Quality. SSSA Publ. No. 49. Soil Science Society of America., 677 South Segoe Rd., Madison, WI 53711, USA.
Evanylo, G.K. and M.M Alley. 1996. Nitrogen soil testing for corn in Virginia. Virginia Coop. Ext. Publ. 418-016.
Evanylo, G.K. and M.M Alley. 1997. Presidedress soil nitrogen test for corn in Virginia. Commun. Soil Sci. & Plant Anal. 28:1285-1301.
Hillel, D. 1982. Introduction to soil physics. Academic Press, San Diego, CA.
Karlen, D.L., M.J. Mausbach, J.W. Doran, R.G. Cline, R.F. Harris, and G.E. Schuman. 1997. Soil quality: A concept, definition, and framework for evaluation. Soil Sci. Soc. Amer. J. 61:4-10.
Keeney D. R. , and D. W. Nelson. 1982. In: A.L. Page (ed.) Methods of Soil Analysis, Part 2 (Second Edition). Chemical and Microbiological Properties. ASA-SSSA, Agronomy Monograph 9: 643-698.
Klute, A. 1965. In: C.A. Black (ed.) Methods of Soil Analysis, Part 1. Physical and Mineralogical Properties, Including statistics of Measurement and Sampling. ASA-SSSA, Agronomy Monograph 9:273-278.
Lowery, B., M.A. Arshad, R. Lal, and W.J. Hickey. 1996. Soil water parameters and soil quality. p.143-157. In: J.W. Doran and A.J. Jones (eds.) Methods for assessing soil quality. Soil Sci. Soc. Am. Spec. Publ. 49. SSSA, Madison, WI.
McLean, E. O. 1982. In: A.L. Page (ed.) Methods of Soil Analysis, Part 2 (Second Edition). Chemical and Microbiological Properties. ASA-SSSA, Agronomy Monograph 9:199-224.
Sarrantonio, M., J.W. Doran, M.A. Liebig, and J.J. Halvorson. 1996. On-farm assessment of soil quality and health. p.83-106. In: J.W. Doran and A.J. Jones (eds.) Methods for assessing soil quality. SSSA Spec. Publ. 49. Soil Science Society of America, Inc., Madison, Wisconsin, USA.
Woods End Research. 1997. Guide to Solvita testing and managing your soil. Woods End Research Laboratory, Inc., Mt. Vernon, ME.